ProMed Pharma is a leading contract manufacturer of polymer-based drug releasing molded dosage forms and combination device components. Working with both established and early-stage medical device and pharmaceutical companies, ProMed develops robust manufacturing processes and platforms for extended drug release from a variety of materials, including silicones, degradable, and nondegradablethermoplastics.
Implantable drug delivery devices offer several advantages over conventional oral or parenteral dosage forms. First, implantable medical devices allow site specific drug administration where the drug is most needed and may also allow for significantly lower doses of drug to minimize potential side effects. Second, implantable combination devices allow for sustained release of a therapeutic agent. Most importantly, patient compliance increases as the treatment regimen associated with an implantable medical device is generally less burdensome than pills or injections.
Learn More by Visiting Our Pharma Site:
Common examples of drug delivery/combination device applications include:
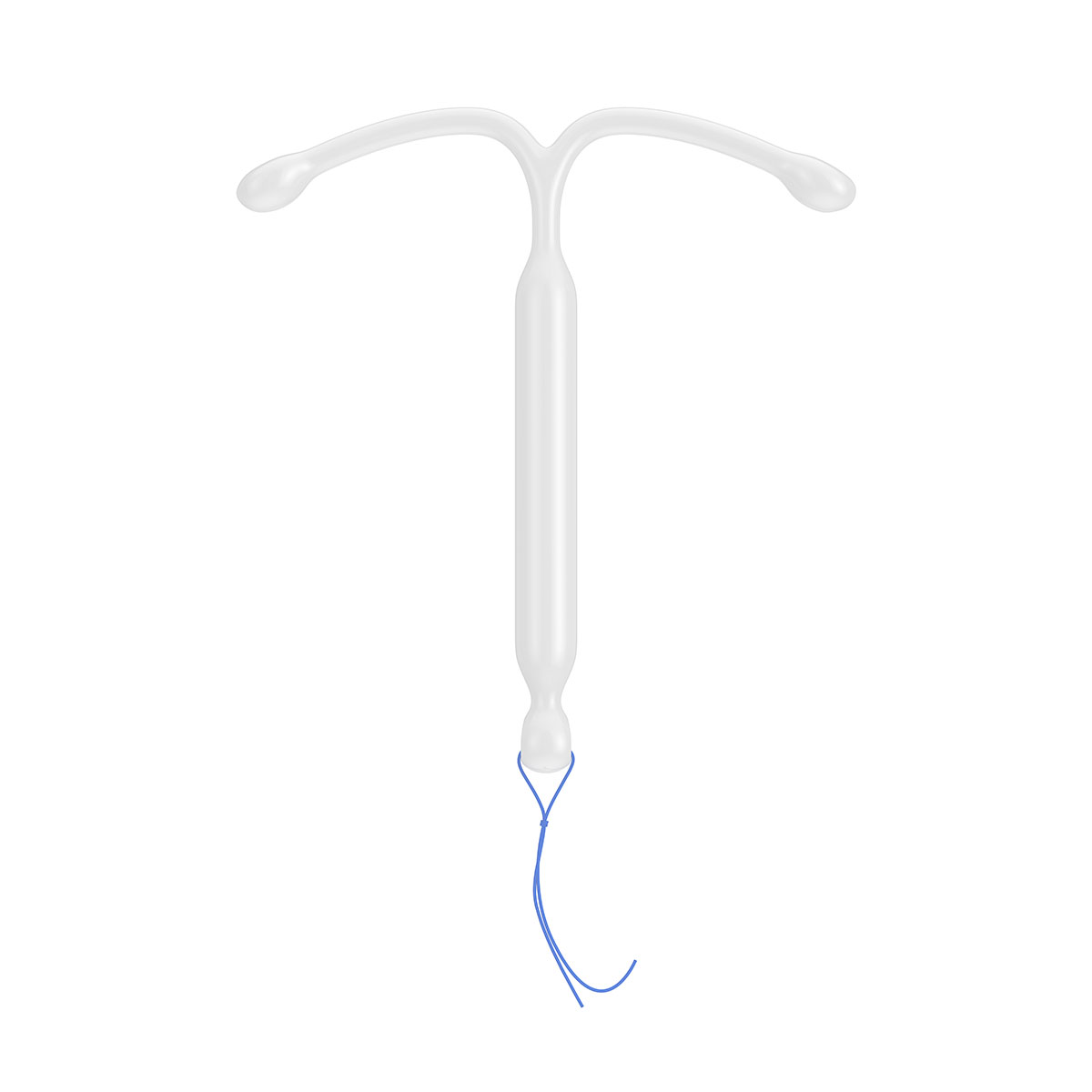
- Women’s Health: Novel drug delivery forms such as subcutaneous implants, intrauterine devices (IUDs) and intravaginal rings (IVRs) are finding increasing applications within the area of women’s health
- Cardiovascular/Neurological: Steroid-eluting pacing and defibrillation leads, paddle leads and anchors are commonly leveraged to mitigate inflammation or infection after device implantation
- Ophthalmology: A number of promising solid implant applications can also be found in ophthalmology for the treatment of back of the eye diseases using steroid to treat glaucoma (prostaglandins) and age-related macular degeneration (anti-VEGF)
- Orthopedic: Steroid eluting spinal implants, hip and joint spacers
- Urological: Antimicrobial catheters, and API’s that specifically target bladder diseases and UTI’s
- Auditory: Steroid eluting coatings and components to mitigate inflammation after device implantation.